Microchiroptera
Echolocating Bats
Nancy B. Simmons and Tenley Conway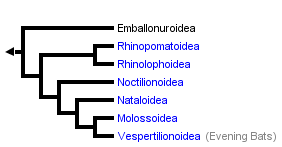

This tree diagram shows the relationships between several groups of organisms.
The root of the current tree connects the organisms featured in this tree to their containing group and the rest of the Tree of Life. The basal branching point in the tree represents the ancestor of the other groups in the tree. This ancestor diversified over time into several descendent subgroups, which are represented as internal nodes and terminal taxa to the right.

You can click on the root to travel down the Tree of Life all the way to the root of all Life, and you can click on the names of descendent subgroups to travel up the Tree of Life all the way to individual species.
For more information on ToL tree formatting, please see Interpreting the Tree or Classification. To learn more about phylogenetic trees, please visit our Phylogenetic Biology pages.
close boxIntroduction
Microchiropteran bats are also known as "echolocating bats" because they have the ability to use echolocation in obstacle avoidance and hunting. These bats occur on every continent but Antartica. Every food preference that is found in bats is represented in Microchiroptera, although the majority are predominatly insectivorous. Several notable microchiropteran bats include Craseonycteris (Bumblebee Bat), which is one of the smallest mammals in the world, Thyroptera (Disc-winged Bats), which attach to leaves using suction cuplike discs on the foot and thumb, and Desmodontinae, which are the only blood-feeding bats.
Classification outlines the higher-level classifications within Microchiroptera.
Echolocation
Microchiropterans navigate with the aid of echolocation, also known as "biosonar." This is similar to the mechanical sonar systems used by humans. A signal is emitted and the returning sound is analyzed to learn about the surrounding environment. Microchiropteran bats are not the only animals that use echolocation. Toothed whales, some insectivores (eg., shrews), oilbirds, and some swiftlets also use echolocation.
Echolocating bats typically emit an ultrasonic (over 15 kilohertz) pulse, and analyze the returning echo to determine the distance to the object as well as what type of object it is (Fenton, 1992). Most bats alternate between emitting sound and listening for returning sound. The frequency, length of call, intensity, and degree of modulations of the emitted sound differs between species, and there may even be differences between individuals within a species.
Echolocation calls are vocalizations that are produced in the larynx (voice box). Calls are emitted through the mouth or the nostrils. Bats that emit calls through the nostrils, such as Phyllostomidae and Rhinolophidae, often have complex folds and/or flaps surrounding the nostrils, which may affect the signal.
A few megachiropterans also use echolocation (e.g., some species of Rousettus), but these bats produce sound by clicking their tongues rather than by vocalization. The different method of sound creation is one reason why echolocation is believed to have evolved independently in Microchiroptera and the few echolocating megachiropteran bats. Echolocation is not thought to have been present in the common ancestor of Megachiroptera and Microchiroptera (Simmons and Geisler, 1998).
The ability to echolocate has allowed many bats to exploit flying nocturnal insects as a food source, as well as to live in dark caves. In neither situation can one successfully rely on vision alone to locate objects due to the limited amount of light. Most likely as a result of increased reliance on echolocation, microchiropterans have reduced vision capabilities, having lost some of the complexity found in the eyes and brains of megachiropteran bats.
While echolocation has many benefits, it also has costs. The most pronounced is that other animals can often hear the signals emitted by bats. Those who are able to hear the sounds include other bats, potential predators, and prey. Some moths have evolved complex ears, apparently for listening to bats. When such a moth hears calls of an approaching bat, it begins evasive maneuvers. Some insects actually emit sounds in response to bat calls. This apparently confuses the bat although it does not directly jam the signal.
Characteristics
Microchiroptera is diagnosed by the following synapomorphies: a free premaxilla, only two lower premolars on each side of the jaw, and a xiphisternum with a medial keel (Simmons and Geisler, 1998). However, these features are troublesome as many lineages within Microchiroptera have apparently secondarily evolved different conditions.
Several other soft-tissue characteristics are found in Microchiroptera, but not Megachiroptera. These characteristics include the presence of a tragus, a small or absent aquaeductus cochleae, the presence of a sophisticated echolocation system, the m. styloglossus originating from the ventral surface of the midpoint of the stylohyal, the clavicle articulating with the coracoid process of the scapula, the m. spinotrapesius clearly differentiated from the trapezius complex, the m. acromiodeltoideus not originating from the thoracic vertebra, the angle of the spinal cord between the dorsal horns from 0 to 25°, and the inferior colliculus larger than the superior colliculus (Simmons and Geisler, 1998). As these features can not be examined in the fossil material collected to date, it is not known whether they also occured in fossils which are closely related to Microchiroptera. Thus, these characteristics can only indirectly support microchiropteran monophyly.
Discussion of Phylogenetic Relationships
Monophyly of Microchiroptera is strongly supported. A variety of data have been analyzed to help resolve the relationships of families within this clade, including osteological characteristics (Smith, 1976; Van Valen, 1979), fetal membrane traits (Luckett, 1980), auditory region morphology (Novacek, 1980), hyoid region characteristics (Griffiths and Smith, 1991; Griffiths et al., 1992), transferrin immunological distance data (Pierson, 1986), rDNA restriction sites (Baker et al., 1991), and DNA hybridization data (Kirsch et al., In press). The most recent comprehensive analyses were those of Simmons (1998) and Simmons and Geisler (1998), who combined all of these data (with the exception of immunological and DNA-hybridization data). Analysis of this large data set produced a relatively well-resolved, well supported phylogeny of Microchiroptera. Interestingly, this tree is largely compatible with trees produced from distance data by Kirsch et al. (In press).
An early phylogentic hypothesis proposed by Smith (1976) indicated two clades within Microchiroptera: Yinochiroptera (Emballonuridae, Rhinolopoidea) and Yangochiroptera (Noctilionoidea and Vespertilionoidea). Other analyses, however, have not supported these clades. Van Valen’s (1979) tree suggests that neither Yinochiroptera nor Yangochiroptera are monophyletic due to a close relationship between Emballonuridae and Noctilionoidea. Pierson (1986) concluded that Yinochiroptera was monophyletic, but due to the close relationship between Vespertilioninae and Yinochiroptera, Yangochiroptera was not. Simmons (1998) and Simmons and Geisler’s (1998) analyses suggested the reverse, a monophyletic Yangochiroptera and a paraphyletic Yinochiroptera. The latter appears paraphyletic due to Emballonuridae placed as the basal branch in Microchiroptera.
Alternative Trees:
Smith (1976)
==== Emballonuridae
==Yinochiroptera=|
| ==== Rhinoloppoidea
=====|
| === Noctilionoidea (Phyllostomoidea)
==Yangochiroptera=|
=== Vespertilionoidea
Van Valen (1979)
============ Rhinopomatidae
|
| ========= Rhinolophoidea
| |
===| | === Emballonuridae
| ===| ===|
=====| ===| === Craseonycteridae
| |
| ====== Noctilionoidea
|
| ============ Vespertilionoidea (including Mystacinidae)
===|
============ Natalidae (including Thyropteridae and Natalidae)
Pierson (1986)
========= Emballonuridae
|
| === Rhinolophidae
| ===|
===| | === Hipposideridae
===| ===|
===| | | === Megadermatidae
===| | | ===|
===| | | | === Rhinopomatidae
=====| | | | |
| | | | ============ vespertilionids
| | | |
| | | =============== Molossidae
| | |
| | ================== Thyropteridae
| |
| ===================== Noctilionoidea (including Mystacinidae)
|
======================== Natalidae
The placement of Emballonuridae has been often debated. Smith placed Emballonuridae in a monophyletic group containing Craseonycteridae and Rhinopomatidae, while Van Valen’s analysis indicated that Emballonuridae and Craseonycteridae were more closely related to Noctilionoidea and Rhinolophoidea than to Rhinopomatidae. Pierson’s tree also indicated that Rhinopomatidae belongs in a clade with Rhinolophoidea, not grouped with Emballonuridae, and did not support a close relationship between Emballonuridae and Noctilionoidea.
The relationships of members of the Vespertilionoidea have long been unclear. Vespertilionidae has been treated as a clade by most authors (Smith, 1976; Van Valen, 1979; Novacek, 1991). However, the frequent appearance of this group in discussions of bats may be attributed to the common assumption of monophyly rather than strong phylogenetic support for this hypothesis. Pierson (1986), who did not assume Vespertilionidae monophyly, placed Tomopeatinae and Miniopterinae (usually found within Vespertilionidae) with Molossoidea and suggested the remaining vespertilionids form a clade with Yinochiroptera. Recent work (Simmons, 1998; Simmons and Geisler, 1998) has indicated that Vespertilionidae as traditionally recognized is paraphyletic. Instead, Antrozoinae (which Simmons [1998] raised from subfamily to family rank) appears closely related to Molossidae and should therefore be placed in Molossoidea; Myzopodidae, Thyropteridae, Fruipteridae and Natalidae represent a clade and are here represented in the superfamily Nataloidea. With the removal of the preceding families, Vespertilionidae, limited to Vespertilioninae, Miniopterinae, Myotinae, Murininae and Kerivoulinae, is now monophyletic." One group which is fairly well supported is the clade containing Phyllostomidae, Noctilionidae, and Mormoopidae (= Noctilionoidea). Mystacinidae, traditionally linked with vespertilionids and molossids, now appears to be closely related to this group (Pierson, 1986; Kirsch et al., In press) and is therfore here included in the Noctilionoidea.
References
Altringham, J. D. 1996. Bats: biology and behavior. New York: Oxford University Press.
Fenton, M. B. 1992. Bats. New York: Facts on File, Inc.
Fenton, M. B. 1995. Natural History and Biosonar Signals.In Hearing by Bats. A.N. Popper and R.R. Fay (eds.). New York: Springer-Verlag.
Griffiths, T. A., and A. L. Smith. 1991. Systematics of emballonuroid bats (Chiroptera: Emballonuridae and Rhinpomatidae), based on hyoid morphology. Bulletin American Museum of Natural History, 206:62-83.
Griffiths, T. A., A. Truckenbrod, and P. J. Sponholtz. 1992. Systematics of megadermatid bats (Chriroptera, Megadermatidae), based on hyoid morphology. American Museum Novitates, 3041:1-21.
Grinnell, A. D. 1995. Hearing in bats: an overview. In Hearing by Bats. A.N. Popper and R.R. Fay (eds.). New York: Springer-Verlag.
Henson, O. W., Jr. 1970. The central nervous system. In. Biology of Bats. W.A. Wimsatt (eds.). vol2. Pp 58-152.
Hill, J. E., and J. D. Smith. 1984. Bats: a natural history. Austin: University of Texas Press.
Kirsch, J. A., J. M. Hutcheon, D. G. Byrnes & B. D. Llyod. 1998. Affinites and historical zoogeography of the New Zealand Short0tailed bat, Mystacina tuberculata Gray 1843, inferred from DNA-hybridization comparisons. Journal of Mammalian Evolution 5(1): 33-64.
Luckett, W. P. 1980. The use of fetal membrane characters in assessing chiropteran phylogeny. Pp. 245-266 In Proceedings Fifth International Bat Research Conference. D.E. Wilson and A.L. Gardner (eds.). Lubbock, Texas: Texas Tech Press.
Novacek, M. J. 1980. Phylogenetic analysis of the chiropteran auditory region. Pp. 317-330 In Proceedings Fifth International Bat Research Conference. D.E. Wilson and A.L. Gardner (eds.). Lubbock, Texas: Texas Tech Press.
Novacek, M. J. 1991. Aspects of morphology of the cochlea in microchiropteran bats: an investigation of character transformation. Bulletin American Museum of Natural History, 206:84-100.
Pierson, E. D. 1986. Molecular systematics of the Microchiroptera: higher taxon relationships and biogeography. Ph.D. dissertation, University of California, Berkeley, California.
Simmons, N. B. 1998. A reappraisal of interfamilial relationships of bats. In Bats: Phylogeny, Morphology, Echolocation and Conservation Biology. T.H. Kunz and P.A. Racey (eds.). Washington: Smithsonian Institution Press.
Simmons, N. B. & J. H. Geisler. 1998. Phylogenetic relationships of Icaronycteris, Archeonycteris, Hassianycteris, and Palaeochiropteryx to extant bat lineages, with comments on the evolution of echolocation and foraging strategies in microchiroptera. Bulletin of the American Museum of Natural History, 235:1-82.
Smith, J. D. 1976. Chiropteran evolution. Pp. 49-69 In Biology of bat of the New World family Phyllostomatidae, Part I. R.J. Baker, J.K. Jones, and D.C. Carter (eds.). Special Publications of the Museum. No. 10. Lubbock, Texas: Texas Tech University.
Van Valen, T. A. 1979. The evolution of bats. Evolutionary Theory, 4:104-121.
Wilson, D. E. 1997. Bats in question. Washinton: Smithsonian Institution Press.
Title Illustrations

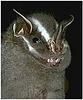
Pteronotus parnellii (Noctilionoidea) and Uroderma bilobatum (Noctilionoidea)
| Scientific Name | Pteronotus parnellii |
|---|---|
| Image Use |
 This media file is licensed under the Creative Commons Attribution-NonCommercial License - Version 3.0. This media file is licensed under the Creative Commons Attribution-NonCommercial License - Version 3.0.
|
| Copyright |
© 1997 Nancy B. Simmons

|
| Scientific Name | Uroderma bilobatum |
|---|---|
| Image Use |
 This media file is licensed under the Creative Commons Attribution-NonCommercial License - Version 3.0. This media file is licensed under the Creative Commons Attribution-NonCommercial License - Version 3.0.
|
| Copyright |
© 1997 Nancy B. Simmons

|
About This Page
Nancy B. Simmons

American Musuem of Natural History, New York, New York, USA
Tenley Conway

University of Toronto at Mississauga, Ontario, Canada
Correspondence regarding this page should be directed to Nancy B. Simmons at
Page copyright © 1997 Nancy B. Simmons
 Page: Tree of Life
Microchiroptera. Echolocating Bats.
Authored by
Nancy B. Simmons and Tenley Conway.
The TEXT of this page is licensed under the
Creative Commons Attribution License - Version 3.0. Note that images and other media
featured on this page are each governed by their own license, and they may or may not be available
for reuse. Click on an image or a media link to access the media data window, which provides the
relevant licensing information. For the general terms and conditions of ToL material reuse and
redistribution, please see the Tree of Life Copyright
Policies.
Page: Tree of Life
Microchiroptera. Echolocating Bats.
Authored by
Nancy B. Simmons and Tenley Conway.
The TEXT of this page is licensed under the
Creative Commons Attribution License - Version 3.0. Note that images and other media
featured on this page are each governed by their own license, and they may or may not be available
for reuse. Click on an image or a media link to access the media data window, which provides the
relevant licensing information. For the general terms and conditions of ToL material reuse and
redistribution, please see the Tree of Life Copyright
Policies.
Citing this page:
Simmons, Nancy B. and Tenley Conway. 1997. Microchiroptera. Echolocating Bats. Version 01 January 1997. http://tolweb.org/Microchiroptera/16085/1997.01.01 in The Tree of Life Web Project, http://tolweb.org/








 Go to quick links
Go to quick search
Go to navigation for this section of the ToL site
Go to detailed links for the ToL site
Go to quick links
Go to quick search
Go to navigation for this section of the ToL site
Go to detailed links for the ToL site